分子别名(Synonym)
G-CSF,CSF3,C17orf33,CSF3OS,MGC45931
表达区间及表达系统(Source)
Human G-CSF, premium grade (GCF-H5214) is expressed from human 293 cells (HEK293). It contains AA Thr 31 - Pro 204 (Accession # NP_757373.1).
Predicted N-terminus: Thr 31
It is produced under our rigorous quality control system that incorporates a comprehensive set of tests including sterility and endotoxin tests. Product performance is carefully validated and tested for compatibility for cell culture use or any other applications in the early preclinical stage. When ready to transition into later clinical phases, we also offer a custom GMP protein service that tailors to your needs. We will work with you to customize and develop a GMP-grade product in accordance with your requests that also meets the requirements for raw and ancillary materials use in cell manufacturing of cell-based therapies.
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries no "tag".
The protein has a calculated MW of 18.7 kDa. The protein migrates as 18 kDa under reducing (R) condition (SDS-PAGE).
内毒素(Endotoxin)
Less than 0.1 EU per μg by the LAL method.
无菌(Sterility)
The sterility testing was performed by membrane filtration method.
支原体(Mycoplasma)
Negative.
纯度(Purity)
>90% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
电泳(SDS-PAGE)

Human G-CSF, premium grade on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 90%.
活性(Bioactivity)-ELISA
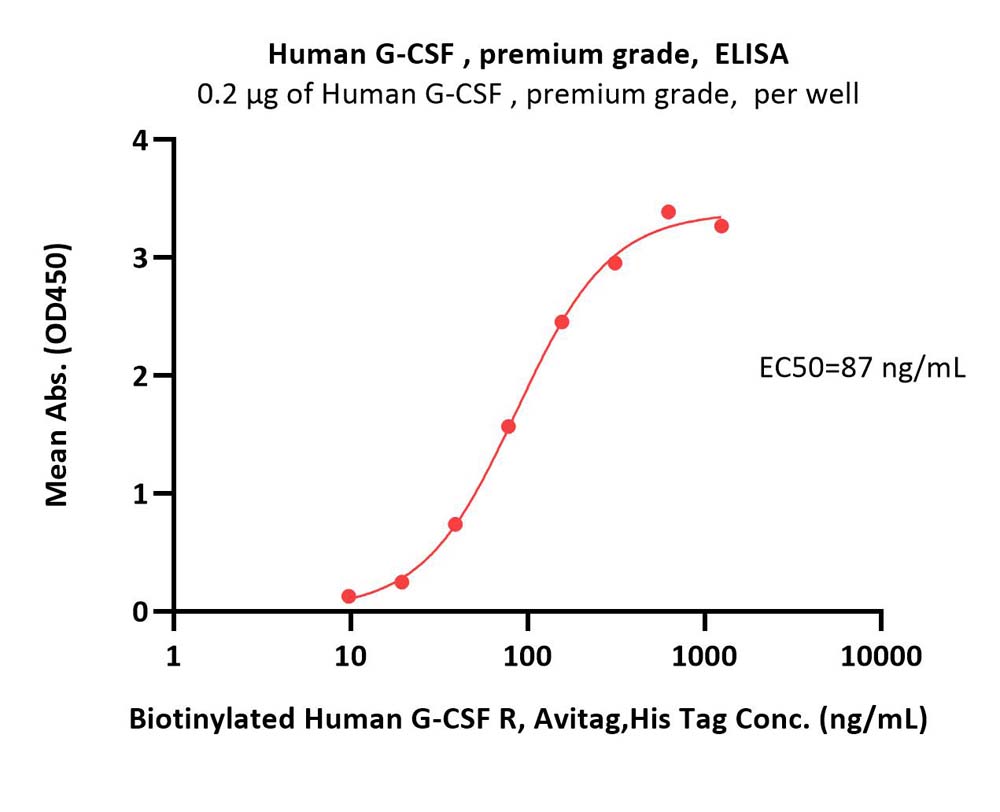
Immobilized Human G-CSF, premium grade (Cat. No. GCF-H5214) at 2 μg/mL (100 μL/well) can bind Biotinylated Human G-CSF R, Avitag,His Tag (Cat. No. GCR-H82E4) with a linear range of 10-156 ng/mL (QC tested).
Protocol
背景(Background)
Granulocyte colony-stimulating factor (G-CSF or GCSF) is also known as colony-stimulating factor 3, CSF3, C17orf33, CSF3OS, GCSF, MGC45931. It is a glycoprotein, growth factor and cytokine produced by a number of different tissues to stimulate the bone marrow to produce granulocytes and stem cells. G-CSF then stimulates the bone marrow to release them into the blood. G-CSF also stimulates the survival, proliferation, differentiation, and function of neutrophil precursors and mature neutrophils. G-CSF regulates them using Janus kinase (JAK)/signal transducer and activator of transcription (STAT) and Ras /mitogen-activated protein kinase (MAPK) and phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt) signal transduction pathway. G-CSF is produced by endothelium, macrophages, and a number of other immune cells. The natural human glycoprotein exists in two forms, a 174- and 180-amino-acid-long protein of molecular weight 19,600 grams per mole. G-CSF can effect on the hematopoietic system and neuronal cells as a neurotrophic factor. The action of G-CSF in the central nervous system is to induce neurogenesis, to increase the neuroplasticity and to counteract apoptosis. G-CSF stimulates the production of white blood cells (WBC). In oncology and hematology, a recombinant form of G-CSF is used with certain cancer patients to accelerate recovery from neutropenia after chemotherapy, allowing higher-intensity treatment regimens. Another form of recombinant human G-CSF called lenograstim is synthesised in Chinese Hamster Ovary cells (CHO cells). The recombinant human G-CSF synthesised in an E. coli expression system is called filgrastim.






















































 膜杰作
膜杰作 Star Staining
Star Staining












