抗体来源(Source)
Anti-SARS-CoV-2 RBD Potent Neutralizing Antibody, Chimeric mAb, Human IgG1 (AM128) (SPD-M128) is a chimeric monoclonal antibody combining the constant domains of the human IgG1 molecule with mouse variable regions. Pseudovirus assay shows that this antibody has potent neutralizing activity against pseudovirus bearing SARS-CoV-2 Spike protein.
克隆号(Clone)
AM128
亚型(Isotype)
Human IgG1 | Kappa
偶联(Conjugate)
Unconjugated
种属反应性(Reactivity)
Virus
特异性(Specificity)
This product is a specific antibody against SARS-CoV-2 Spike protein RBD domain. No cross-reactivity is detected with Spike protein RBD domain of other coronaviruses, including SARS-CoV, MERS-CoV, HCoV-229E, HCoV-NL63, HCoV-OC43 and HCoV-HKU1.
应用(Application)
| Application | Recommended Usage |
| ELISA | 0.1-25 ng/mL |
纯度(Purity)
>95% as determined by SDS-PAGE.
纯化(Purification)
Protein A purified / Protein G purified
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
质量管理控制体系(QMS)
电泳(SDS-PAGE)

Anti-SARS-CoV-2 RBD Potent Neutralizing Antibody, Chimeric mAb, Human IgG1 (AM128) on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95%.
活性(Bioactivity)-ELISA
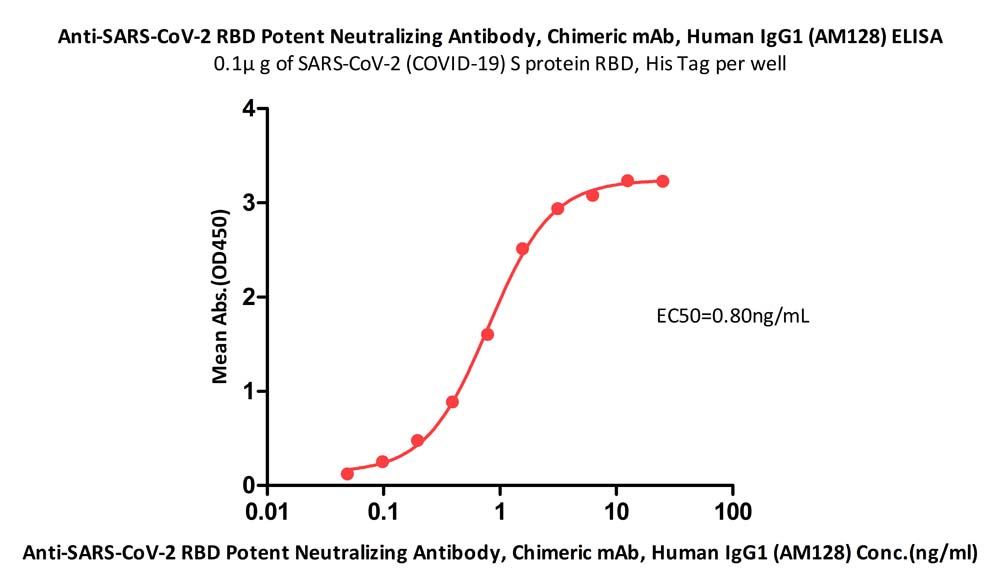
Immobilized SARS-CoV-2 S protein RBD, His Tag (Cat. No. SPD-C52H3) at 1 μg/mL (100 μL/well) can bind Anti-SARS-CoV-2 RBD Potent Neutralizing Antibody, Chimeric mAb, Human IgG1 (AM128) (Cat. No. SPD-M128) with a linear range of 0.05-0.78 ng/mL (QC tested).
Protocol
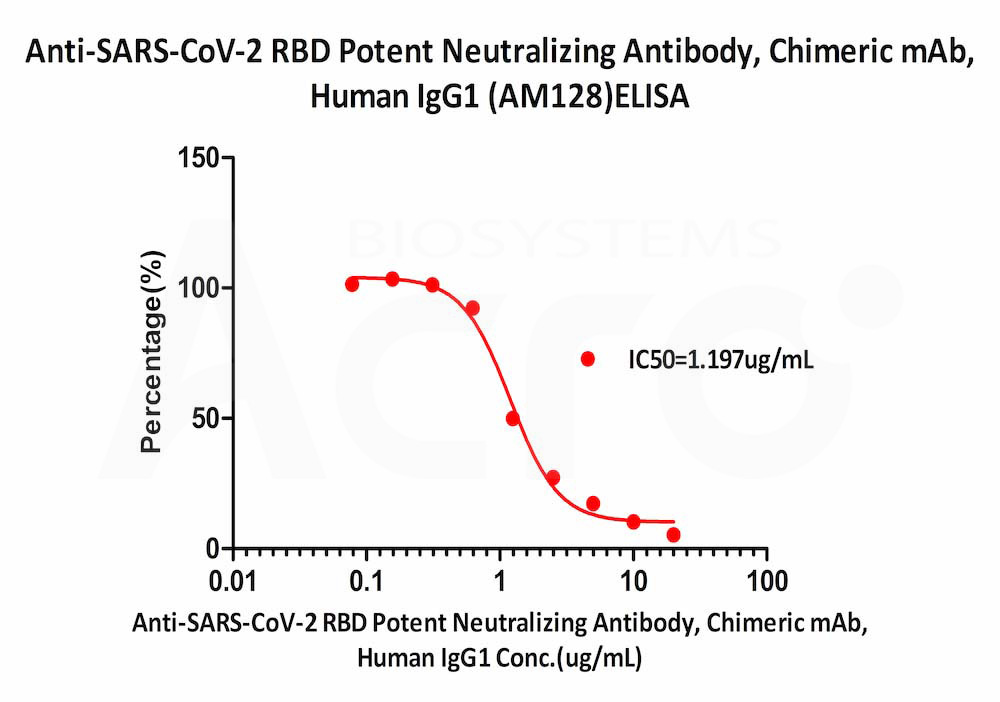
Serial dilutions of Anti-SARS-CoV-2 RBD Potent Neutralizing Antibody, Chimeric mAb, Human IgG1 (AM128) (Cat. No. SPD-M128) was detected by SARS-CoV-2 Inhibitor screening Kit with a half maximal inhibitory concentration (IC50) of 1.197 μg/mL (Routinely tested).
Protocol
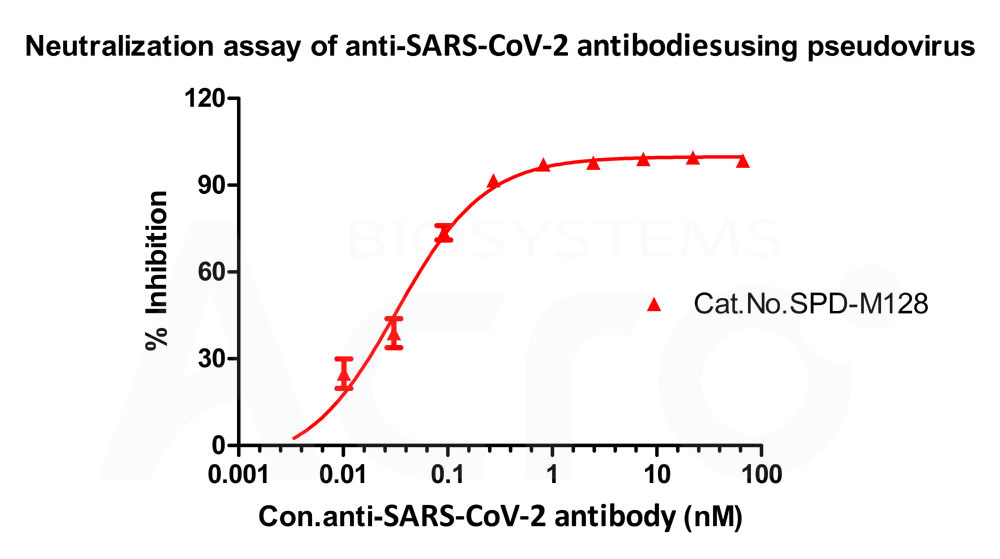
Serial dilutions of Anti-SARS-CoV-2 RBD Potent Neutralizing Antibody (Cat. No. SPD-M128) was incubated with SARS-CoV-2 pseudotyped virus at 37 °C for 1 hour. Afterward, Huh-7 cells were added, followed by incubation at 37 °C for 24h. Chemiluminescence detection was performed and the virus neutralization titers (IC50) is 0.03255 nM using the Reed-Muench method.
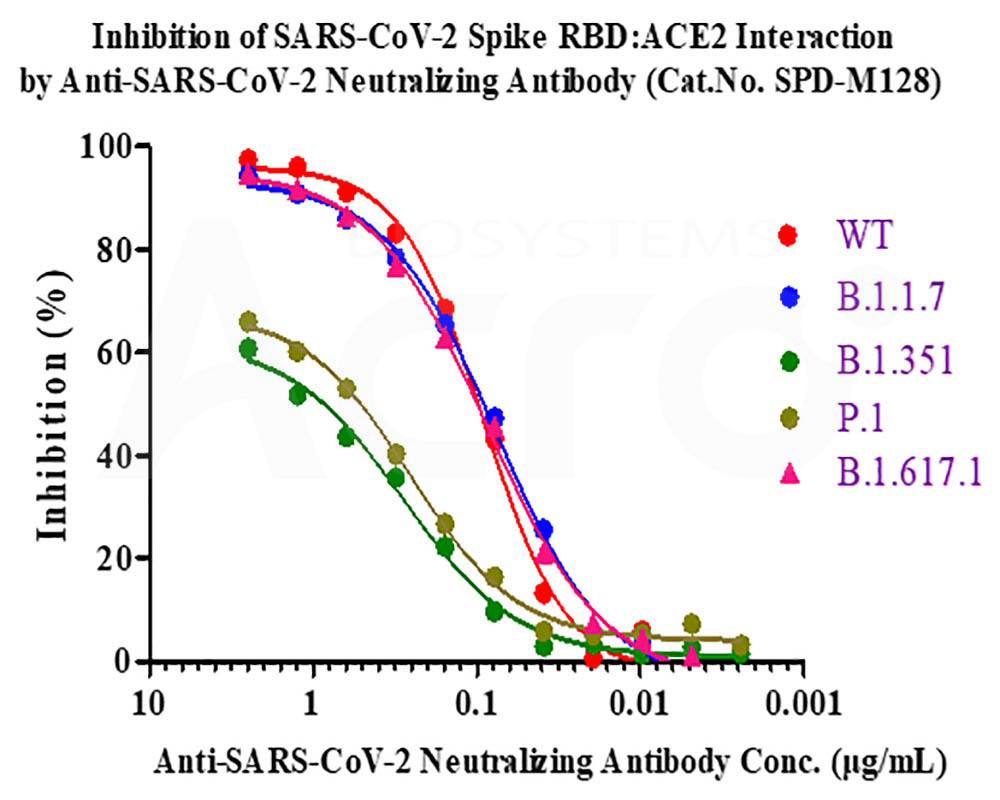
Anti-SARS-CoV-2 Spike RBD Neutralizing antibody (Cat.No. SPD-M128) neutralizes SARS-CoV-2 Spike RBD by inhibiting RBD:ACE2 interaction. The ACE2-coated plate is incubated with the wild type (WT) RBD or B.1.1.7, B.1.351, P.1, B.1.617.1 mutant and treated with the antibody at increasing concentration. Percent inhibition is calculated based on the OD value.
Protocol
活性(Bioactivity)-BLI
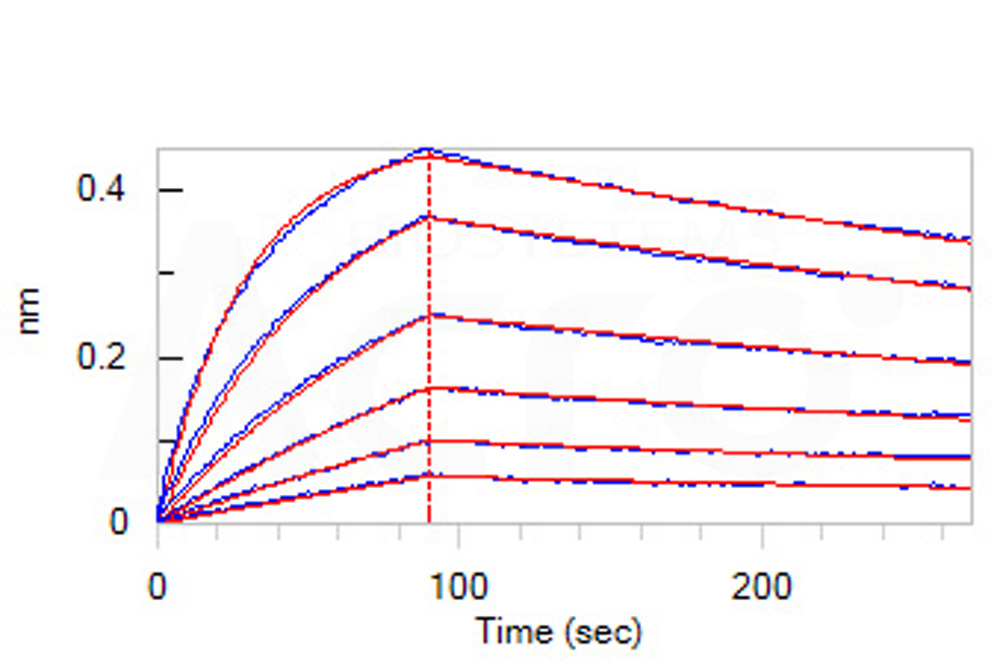
Loaded Anti-SARS-CoV-2 RBD Potent Neutralizing Antibody, Chimeric mAb, Human IgG1 (AM128) (Cat. No. SPD-M128) on AHC Biosensor, can bind SARS-CoV-2 S1 protein, His Tag with an affinity constant of 4.42 nM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol
 +添加评论
+添加评论背景(Background)
It's been reported that Coronavirus can infect the human respiratory epithelial cells through interaction with the human ACE2 receptor. The spike protein is a large type I transmembrane protein containing two subunits, S1 and S2. S1 mainly contains a receptor binding domain (RBD), which is responsible for recognizing the cell surface receptor. S2 contains basic elements needed for the membrane fusion.The S protein plays key parts in the induction of neutralizing-antibody and T-cell responses, as well as protective immunity.























































 膜杰作
膜杰作 Star Staining
Star Staining
















