The deLIVERed promises of gene therapy: past, present and future of liver-directed gene therapyPuzzo, Kay
Mol Ther (2025)
Abstract: Gene therapy has revolutionized modern medicine by offering innovative treatments for genetic and acquired diseases. The liver, has and continues as a prime target for in vivo gene therapy due to its essential biological functions, vascular access to the major target cell (hepatocytes) and relatively immunotolerant environment. Adeno-associated virus (AAV) vectors have become the cornerstone of liver-directed therapies, demonstrating remarkable success in conditions such as hemophilia A and B, with FDA approved therapies like Etranacogene Dezaparvovec, Beqvez, and Roctavian marking milestones in the field. Despite these advances, challenges persist, including vector immunogenicity, species-specific barriers, and high manufacturing costs. Innovative strategies, such as capsid engineering, immune modulation, and novel delivery systems are continuing to address these issues in expanding the scope of therapeutic applications. Some of the challenges with many new therapies result in the discordance between preclinical success and translation into humans. The advent of various genome-editing tools to repair genomic mutations or insert therapeutic DNAs into precise locations in the genome further enhances the potential for a single-dose medicine that will offer durable life-long therapeutic treatments. As advancements accelerate, liver-targeted gene therapy is poised to continue to transform the treatment landscape for both genetic and acquired disorders, for which unmet challenges remain.Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
The curious case of AAV immunologyKeeler, Zhan, Ram
et alMol Ther (2025)
Abstract: Immune responses to adeno-associated virus (AAV) have long been perplexing, from its first discovery to the latest clinical trials of recombinant AAV (rAAV) therapy. Wild-type AAV (wtAAV) does not cause any known disease, making it an ideal vector for gene therapy, as viral vectors retain virus-like properties. Although AAV stimulates only a mild immune response compared to other viruses, it is still recognized by the innate immune system and induces adaptive immune responses. B cell responses against both wtAAV and rAAV are robust and can hinder gene therapy applications and prevent redosing. T-cell responses can clear transduced cells or establish tolerance against gene therapy. Immune responses to AAV gene therapy are influenced by many factors. Most clinical immunotoxicities that develop in response to gene therapies have emerged as higher doses of AAV vectors have been utilized and were not properly modeled in pre-clinical animal studies. Thus, several strategies have been undertaken to reduce or mitigate immune responses to AAV. While we have learned a considerable amount about how the immune system responds to AAV gene therapy since the discovery of AAV virus, it still remains a curious case that requires more investigation to fully understand.Copyright © 2025. Published by Elsevier Inc.
Gene Therapy for Hemophilia - From Basic Science to First Approvals of "One-and-Done" TherapiesHerzog, Kaczmarek, High
Mol Ther (2025)
Abstract: Realistic paths to gene therapy for the X-linked bleeding disorder hemophilia started to materialize in the mid 1990s, resulting in disease correction in small and large animal models. Out of a diversity of approaches, in vivo adeno-associated viral (AAV) gene transfer to hepatocytes emerged as the most promising strategy, eventually forming the basis for multiple advanced clinical trials and regulatory approval of two products for the treatment of hemophilia B (coagulation factor IX deficiency) and one for hemophilia A (factor VIII deficiency). Ideally, gene therapy is effective with a single administration, thus providing therapeutic factor levels over a period of years, without the need for frequent injections. Overcoming multiple obstacles, some not predicted by pre-clinical studies, sustained partial to complete correction of coagulation for several years to an entire decade has now been documented in patients, with observation ongoing. A hyperactive form of FIX improved efficacy in hemophilia B, and superior engineered variants of FVIII are emerging. Nonetheless, challenges remain, including pre-existing immunity to AAV capsids, toxicities, inter-patient variability in response to treatment, and difficulty in obtaining durable therapeutic expression of FVIII. In alternative approaches, in vivo gene editing and ex vivo gene therapies targeting hemopoietic cells are in development.Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
The dual role of titanium particles in osteolysis: implications for gene therapy in prosthesis looseningChen, Guo, Wu
et alEur J Med Res (2025) 30 (1), 216
Abstract: Aseptic prosthesis loosening caused by wear particles is a major complication in patients with osteoarthritis following total joint replacement. Despite advancements in treatment, the underlying mechanisms remain poorly understood, and effective therapies are still lacking.In this study, we investigated the effects of titanium particles on osteoclast and osteoblast differentiation through both in vitro and in vivo experiments.Our findings revealed that titanium particles not only promote the differentiation of RAW264.7 cells into osteoclasts and enhance the secretion of inflammatory factors but also inhibit the differentiation of BMSCs into osteoblasts and reduce the expression of Wnt signaling pathway-related factors. Furthermore, using a mouse model of knee prosthesis loosening and AAV-mediated gene therapy, we demonstrated that the combination of TNF-α interference and Wnt3a overexpression was more effective than single-gene therapy in reducing inflammatory cell infiltration, promoting bone formation, and mitigating bone destruction.These results highlight the dual role of titanium particles in periprosthetic osteolysis and underscore the potential of a gene therapy strategy targeting both inflammatory factors and the Wnt signaling pathway to improve knee prosthesis loosening. This study provides a scientific foundation for developing novel therapeutic approaches.© 2025. The Author(s).
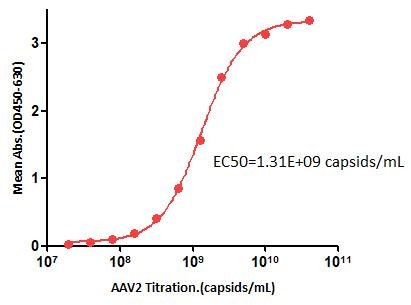
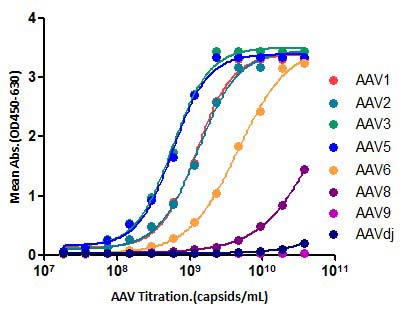























































 膜杰作
膜杰作 Star Staining
Star Staining















