分子别名(Synonym)
IL-17F,Interleukin-17F,Cytokine ML-1
表达区间及表达系统(Source)
Human IL-17F (H161R), His Tag (ILF-H4240) is expressed from human 293 cells (HEK293). It contains AA Arg 31 - Gln 163 (Accession # Q96PD4-1 (H161R)).
Predicted N-terminus: His
Request for sequence
蛋白结构(Molecular Characterization)

This protein carries a polyhistidine tag at the N-terminus.
The protein has a calculated MW of 16.8 kDa. The protein migrates as 20-23 kDa when calibrated against Star Ribbon Pre-stained Protein Marker under reducing (R) condition (SDS-PAGE) due to glycosylation.
内毒素(Endotoxin)
Less than 1.0 EU per μg by the LAL method.
纯度(Purity)
>95% as determined by SDS-PAGE.
制剂(Formulation)
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
重构方法(Reconstitution)
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
存储(Storage)
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
- -20°C to -70°C for 12 months in lyophilized state;
- -70°C for 3 months under sterile conditions after reconstitution.
质量管理控制体系(QMS)
电泳(SDS-PAGE)
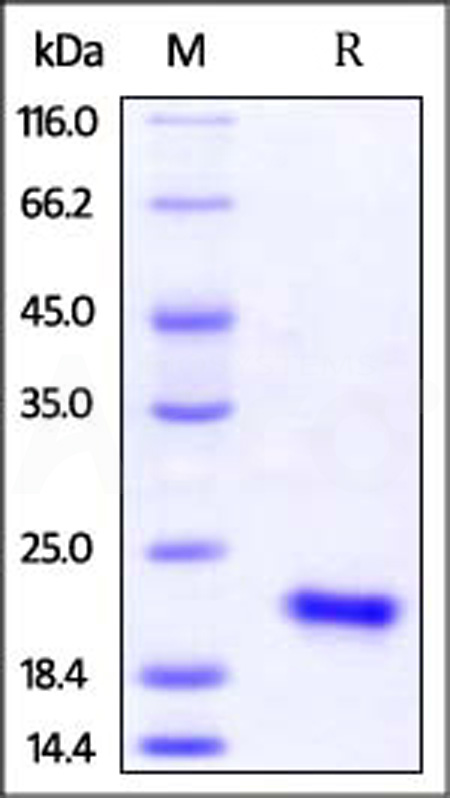
Human IL-17F (H161R), His Tag on SDS-PAGE under reducing (R) condition. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95% (With Star Ribbon Pre-stained Protein Marker).
活性(Bioactivity)-ELISA
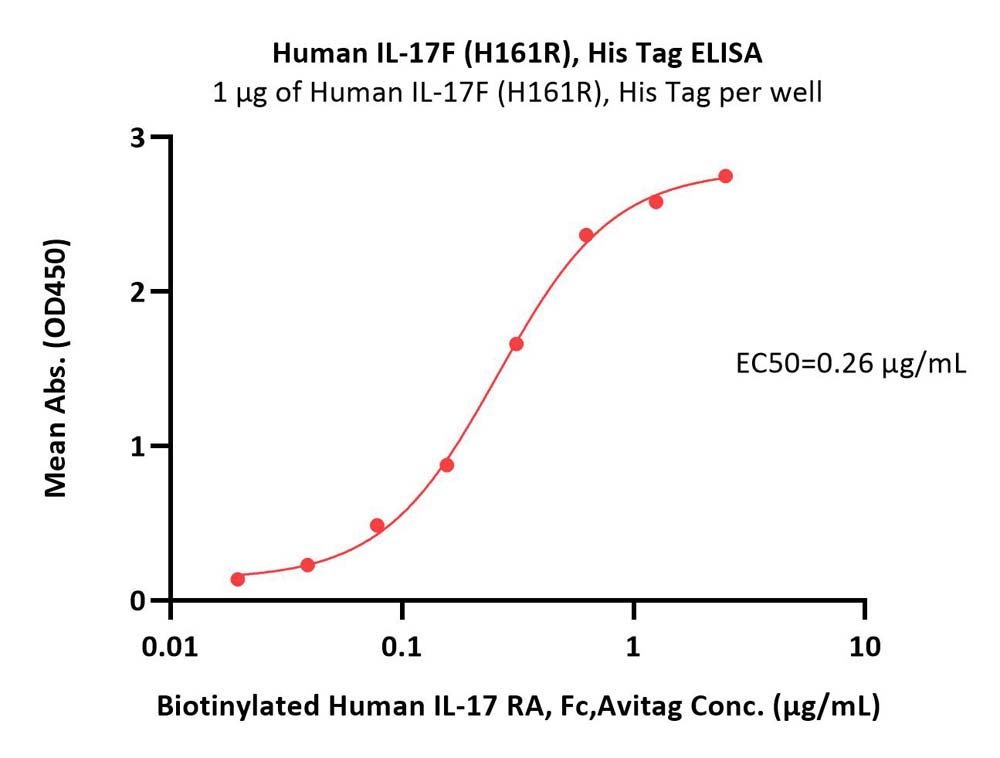
Immobilized Human IL-17F (H161R), His Tag (Cat. No. ILF-H4240) at 10 μg/mL (100 μL/well) can bind Biotinylated Human IL-17 RA, Fc,Avitag (Cat. No. ILA-H82F1) with a linear range of 0.02-0.313 μg/mL (QC tested).
Protocol

Immobilized Human IL-17F (H161R), His Tag (Cat. No. ILF-H4240) at 10 μg/mL (100 μL/well) can bind Human IL-17 RA, Fc Tag with a linear range of 0.078-1.25 μg/mL (Routinely tested).
Protocol

Immobilized Human IL-17F (H161R), His Tag (Cat. No. ILF-H4240) at 2 μg/mL (100 μL/well) can bind Recombinant Human IL-17RC Isoform 1 Fc Chimera with a linear range of 0.2-16 ng/mL (Routinely tested).
Protocol
活性(Bioactivity)-BLI

Loaded Human IL-17 RA, Fc Tag on Protein A Biosensor, can bind Human IL-17F (H161R), His Tag (Cat. No. ILF-H4240) with an affinity constant of 299 nM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol

Loaded Human IL17RA & IL17RC Protein, Fc Tag&Fc Tag (Cat. No. ILC-H5257) on Protein A Biosensor, can bind Human IL-17F (H161R), His Tag (Cat. No. ILF-H4240) with an affinity constant of 1.79 nM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol

Loaded Human IL-17F (H161R), His Tag (Cat. No. ILF-H4240) on NTA Biosensor, can bind Human IL-17 RC, Fc Tag (Cat. No. ILC-H5259) with an affinity constant of 288 nM as determined in BLI assay (ForteBio Octet Red96e) (Routinely tested).
Protocol
背景(Background)
Interleukin-17F (IL17F) is also known as Interleukin-24 (IL24), Cytokine ML-1, is a secreted disulfide-linked homodimer which belongs to the IL-17 family. There are at least six members of the IL-17 family in humans and in mice: IL-17A, IL-17B, IL-17C, IL-17D, IL-17E (also called IL-25) and IL-17F (also called IL-24). IL-17 represents a family of structurally related cytokines that share a highly conserved C-terminal region but differ from one another in their N-terminal regions and in their distinct biological roles. IL-17F / IL-24 is expressed in activated, but not resting, CD4+ T-cells and activated monocytes. IL17F / Interleukin-24 stimulates the production of other cytokines such as IL-6, IL-8 and granulocyte colony-stimulating factor, and can regulate cartilage matrix turnover. IL17F / IL-24 stimulates PBMC and T-cell proliferation and Inhibits angiogenesis. Defects in IL17F are the cause of familial candidiasis type 6 (CANDF6).























































 膜杰作
膜杰作 Star Staining
Star Staining

















