The long noncoding RNA APR attenuates PPRV infection-induced accumulation of intracellular iron to inhibit membrane lipid peroxidation and viral replicationWen, Chang, Yang
et almBio (2025)
Abstract: Peste des petits ruminants virus (PPRV) is an important pathogen that has long been a significant threat to small ruminant productivity worldwide. Iron metabolism is vital to the host and the pathogen. However, the mechanism underlying host-PPRV interactions from the perspective of iron metabolism and iron-mediated membrane lipid peroxidation has not been reported thus far. In this study, we identified a novel host long-noncoding RNA (lncRNA), APR, that impairs PPRV infectivity by sponging miR-3955-5p, a negative microRNA (miRNA) that directly targets the gene encoding the ferritin-heavy chain 1 (FTH1) protein. Importantly, we demonstrated that PPRV infection causes aberrant cellular iron accumulation by increasing transferrin receptor (TFRC) expression and that iron accumulation induces reticulophagy and ferroptosis, which benefits PPRV replication. Moreover, PPRV infection enhanced the localization of cellular iron on the endoplasmic reticulum (ER) and caused ER membrane damage by promoting excess lipid peroxidation to induce reticulophagy. Interestingly, APR decreased PPRV infection-induced accumulation of intracellular Fe2+ via miR-3955-5p/FTH1 axis and ultimately inhibited reticulophagy and ferroptosis. Additionally, our results indicate that interferon regulatory factor 1 promotes APR transcription by positively regulating APR promoter activity after PPRV infection. Taken together, our findings revealed a new pattern of PPRV-host interactions, involving noncoding RNA regulation, iron metabolism, and iron-related membrane lipid peroxidation, which is critical for understanding the host defense against PPRV infection and the pathogenesis of PPRV.IMPORTANCEMany viruses have been demonstrated to engage in iron metabolism to facilitate their replication and pathogenesis. However, the mechanism by which PPRV interacts with host cells from the perspective of iron metabolism, or iron-mediated membrane lipid peroxidation, has not yet been reported. Our data provide the first direct evidence that PPRV infection induces aberrant iron accumulation to promote viral replication and reveal a novel host lncRNA, APR, as a regulator of iron accumulation by promoting FTH1 protein expression. In this study, PPRV infection increased cellular iron accumulation by increasing TFRC expression, and more importantly, iron overload increased viral infectivity as well as promoted ER membrane lipid peroxidation by enhancing the localization of cellular iron on the ER and ultimately induced ferroptosis and reticulophagy. Furthermore, a host factor, the lncRNA APR, was found to decrease cellular iron accumulation by sponging miR-3955-5p, which directly targets the gene encoding the FTH1 protein, thereby attenuating PPRV infection-induced ferroptosis and reticulophagy and inhibiting PPRV infection. Taken together, the results of the present study provide new insight into our understanding of host-PPRV interaction and pathogenesis from the perspective of iron metabolism and reveal potential targets for therapeutics against PPRV infection.
Evaluation of serum levels of calprotectin, lactoferrin and zinc in patients with type II diabetes mellitusHassan, Shamran, Albadry
et alWiad Lek (2025) 78 (2), 288-294
Abstract: Aim: The current study aimed to evaluate the role of serum calprotectin, lactoferrin and serum zinc in patients with type II diabetes mellitus.Materials and Methods: Sixty subjects have been investigated in this study, (40) patients with T2DM and 20 apparently healthy participants (control group) during the period between October 2021 to January 2022. Zinc was measured using a calorimetric method, while calprotectin and lactoferrin were measured by ELISA.Results: The findings also revealed that levels of serum calprotectin and lactoferrin have significantly increased in in patients with T2DM as compared to healthy subjects. The result also showed that serum zin is decreased in patients groups. The correlation matrix exhibited that there was a strong positive correlation between calprotectin and lactoferrin, a significant negative correlation between zinc and calprotectin.Conclusions: It was concluded that high serum calprotectin and lactoferrin indicated a strong inflammatory status in T2DM patients. Zinc is likely to be negative affected by the high inflammatory response indicated by that high serum calprotectin.
Altered chaperone-nonmuscle myosin II interactions drive pathogenicity of the UNC45A c.710T>C variant in osteo-oto-hepato-enteric syndromeWaich, Kreidl, Vodopiutz
et alJCI Insight (2025) 10 (6)
Abstract: The osteo-oto-hepato-enteric (O2HE) syndrome is a severe autosomal recessive disease ascribed to loss-of-function mutations in the Unc-45 myosin chaperone A (UNC45A) gene. The clinical spectrum includes bone fragility, hearing loss, cholestasis, and life-threatening diarrhea associated with microvillus inclusion disease-like enteropathy. Here, we present molecular and functional analysis of the UNC45A c.710T>C (p.Leu237Pro) missense variant, which revealed a unique pathogenicity compared with other genetic variants causing UNC45A deficiency. The UNC45A p.Leu237Pro mutant retained chaperone activity, prevented myosin aggregation, and supported proper nonmuscle myosin II (NMII) filament formation in patient fibroblasts and human osteosarcoma (U2OS) cells. However, the mutant formed atypically stable oligomers and prevented chaperone-myosin complex dissociation, thereby inhibiting NMII functions. Similar to biallelic UNC45A deficiency, this resulted in impaired intracellular trafficking, defective recycling, and abnormal retention of transferrin at various endocytic sites. In particular, coexpression of wild-type protein attenuated the pathogenic effects of the variant by inhibiting excessive oligomer formation. Our results elucidate the pathogenic mechanisms and recessive characteristics of this variant and may aid in the development of targeted therapies.
Wolf in Sheep's Clothing: Taming Cancer's Resistance with Human Serum Albumin?Stukan, Żuk, Pukacka
et alInt J Nanomedicine (2025) 20, 3493-3525
Abstract: Human serum albumin (HSA) has emerged as a promising carrier for nanodrug delivery, offering unique structural properties that can be engineered to overcome key challenges in cancer treatment, especially resistance to chemotherapy. This review focuses on the cellular uptake of albumin-based nanoparticles and the modifications that enhance their ability to bypass resistance mechanisms, particularly multidrug resistance type 1 (MDR1), by improving targeting to cancer cells. In our unique approach, we integrate the chemical properties of albumin, its interactions with cancer cells, and surface modifications of albumin-based delivery systems that enable to bypass resistance mechanisms, particularly those related to MDR1, and precisely target receptors on cancer cells to improve treatment efficacy. We discuss that while well-established albumin receptors such as gp60 and gp18/30 are crucial for cellular uptake and transcytosis, their biology remains underexplored, limiting their translational potential. Additionally, we explore the potential of emerging targets, such as cluster of differentiation 44 (CD44), cluster of differentiation (CD36) and transferrin receptor TfR1, as well as the advantages of using dimeric forms of albumin (dHSA) to further enhance delivery to resistant cancer cells. Drawing from clinical examples, including the success of albumin-bound paclitaxel (Abraxane) and new formulations like Pazenir and Fyarro (for Sirolimus), we identify gaps in current knowledge and propose strategies to optimize albumin-based systems. In conclusion, albumin-based nanoparticles, when tailored with appropriate modifications, have the potential to bypass multidrug resistance and improve the targeting of cancer cells. By enhancing albumin's ability to efficiently deliver therapeutic agents, these carriers represent a promising approach to addressing one of oncology's most persistent challenges, with substantial potential to improve cancer treatment outcomes.© 2025 Stukan et al.

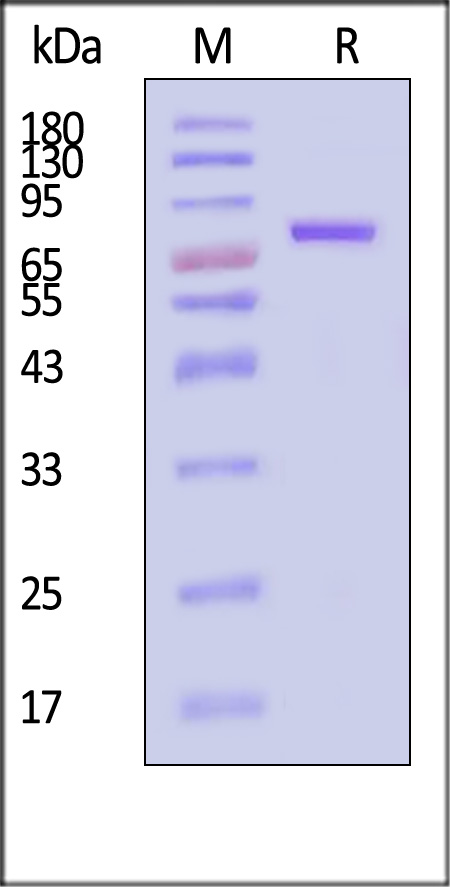
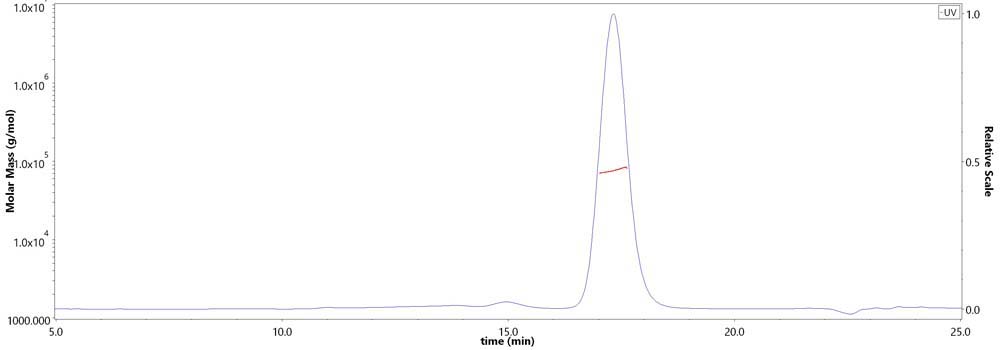
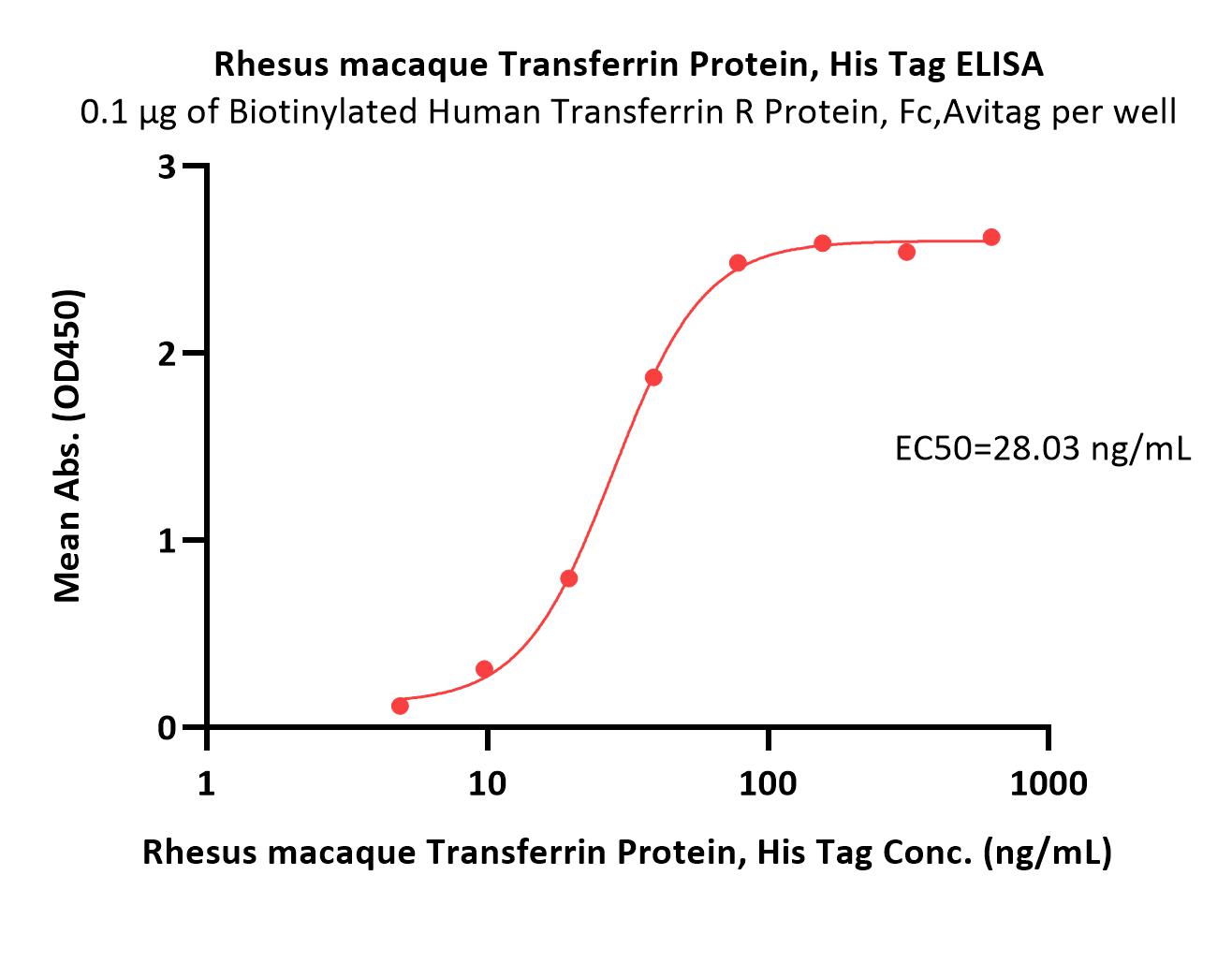























































 膜杰作
膜杰作 Star Staining
Star Staining















